 This is why fusion is still in the research and development phase – and fission is already making electricity. The reasons that have made fusion so difficult to achieve to date are the same ones that make it safe: it is a finely balanced reaction which is very sensitive to the conditions – the reaction will die if the plasma is too cold or too hot, or if there is too much fuel or not enough, or too many contaminants, or if the magnetic fields are not set up just right to control the turbulence of the hot plasma. Unlike nuclear fission, the nuclear fusion reaction in a tokamak is an inherently safe reaction. In conventional nuclear power stations today, there are systems in place to moderate the chain reactions to prevent accident scenarios and stringent security measures to deal with proliferation issues. This chain reaction is the key to fission reactions, but it can lead to a runaway process resulting in nuclear accidents. The result of the instability is the nucleus breaking up, in any one of many different ways, and producing more neutrons, which in turn hit more uranium atoms and make them unstable and so on. Fission reactor systems can be dangerous when poorly designed or managed, as events in Chernobyl and Fukushima have shown. According to the International Atomic Energy Agency (IAEA), it could produce four times. It is triggered by uranium absorbing a neutron, which renders the nucleus unstable. Yes, fusion energy produces a larger amount of energy than fission. Fission and chain reactionsįission is the nuclear process that is currently run in nuclear power plants. Both reactions release energy which, in a power plant, would be used to boil water to drive a steam generator, thus producing electricity. 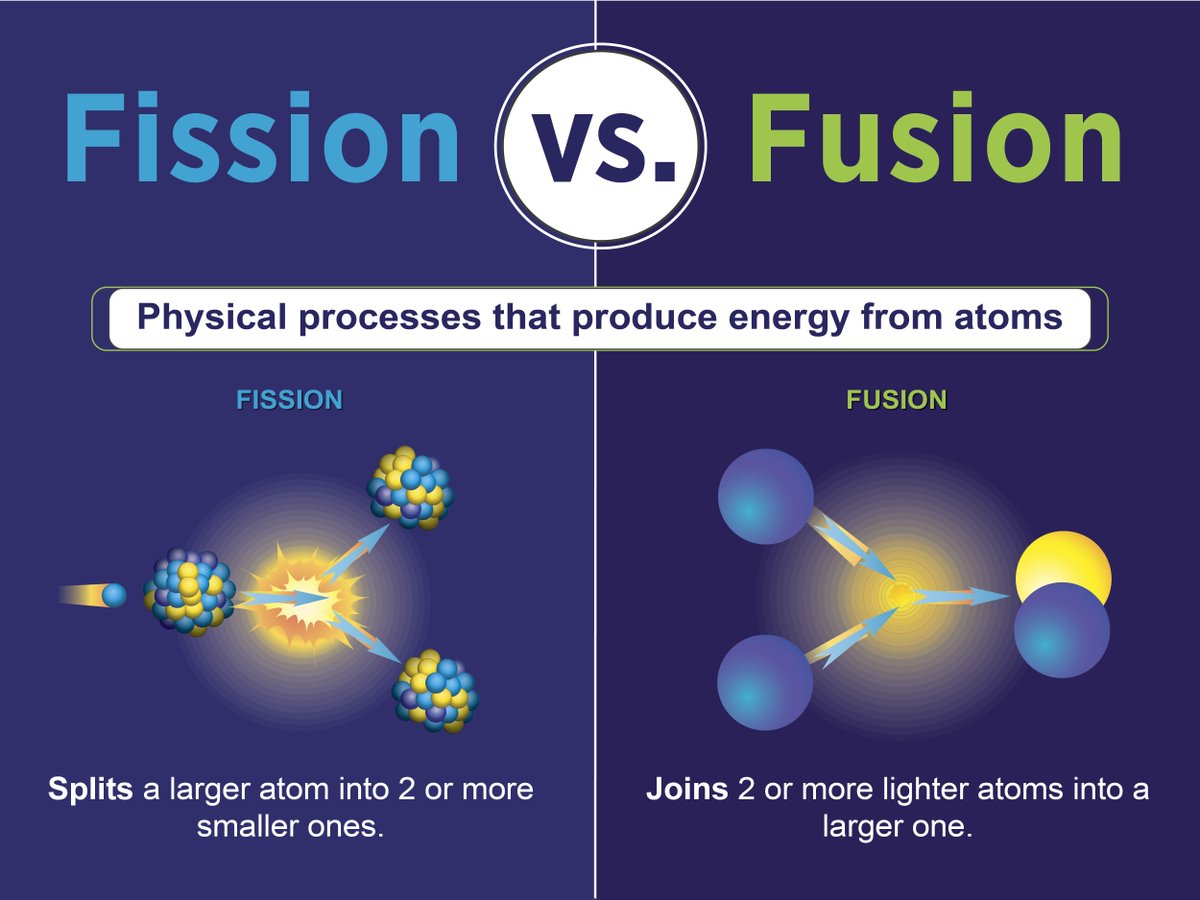
However, fusion is combining light atoms, for example two hydrogen isotopes, deuterium and tritium, to form the heavier helium. There are two methods of doing this: fission and fusion. Releasing this energy would free the world from having to use fossil fuels. In fission, energy is gained by splitting heavy atoms, for example uranium, into smaller atoms such as iodine, caesium, strontium, xenon and barium, to name just a few. The nuclei of atoms contain a large amount of energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
